Featured Image: Fractured sea ice. Image courtesy Pink Floyd 88 a, accessed through Wikimedia Commons GNU Free Documentation License
Paper: Elevated sources of cobalt in the Arctic Ocean
Authors: Randelle Bundy, Alessandro Tagliabue, Nicholas Hawco, Peter Morton, Benjamin Twining, Mariko Hatta, Abigail Noble, Mattia Cape, Seth John, Jay Cullen, Mak Saito
Imagine navigating the Beaufort Sea to the North Pole, crossing icy and treacherous waters through the untamed North, all to chase a metal that is so rare that you have a better chance of finding 5 grains of sand in an Olympic swimming pool*. This is exactly what Bundy et al. accomplished in their work identifying cobalt amounts in the Arctic Ocean and how these amounts vary based on ocean depth, distance from land, and over a time period of 6 years.
In their recent study, they learned that most ocean-bound cobalt actually comes from surrounding land and is deposited into the ocean via rivers. They also found that as climate change causes Arctic temperatures to increase, glaciers, permafrost, and snow are melting at heightened and prolonged rates, depositing this metal into the Arctic Ocean more consistently. From there, a major ocean current will eventually carry the cobalt into the North Atlantic where it becomes available to phytoplankton, potentially stimulating massive growth events capable of altering whole food webs.
In order to capture these metals in the Arctic Ocean, the researchers threw overboard remote- controlled chambers containing specialized bottles capable of opening and closing at designated depths. These water samples were filtered, preserved, and prepared using specially designed molecules that bind only to cobalt. Once these molecules grabbed onto the cobalt, the researchers were able to zap them with electricity and count how many molecules were present and bound to cobalt. There are three main forms of cobalt in the ocean known as “total dissolved,” “particulate,” and “labile,” referring to all cobalt present, the portion of that total that is solid, and the portion that is easily consumed by phytoplankton, respectively. Using these cobalt measurements, the researchers were able to derive and verify an equation that numerically describes the system. Their equation’s accuracy allowed for them to begin predicting future scenarios of cobalt amounts in the Arctic related to time passed, proximity to land, depth in the ocean, and many other climate-related changes that could alter cobalt presence and distribution.
Despite being so rare, cobalt is a key player in the ocean ecosystem, where its scarcity stifles phytoplankton growth. The Arctic Ocean’s chemistry keeps cobalt securely locked away from phytoplankton by encasing it in hard-to-reach microscopic physical structures, coupled with freezing cold temperatures which physically slow down the phytoplankton’s movements. This allows for a buildup of cobalt in the shallow Arctic since there are strict layers in the water column that prevent movement of tiny molecules and creatures out of one layer and into another. Over a 2 to 4-year period, these shallow waters will be shunted by the Transpolar Drift Stream into the Atlantic increasing the amounts of cobalt available to a warmer, more uniform system, where it will be consumed by phytoplankton very quickly. Transport of cobalt into the Atlantic reveals how the tiniest increase of this metal in one part of the world can have far-reaching ecological impacts, much like the butterfly effect.
This biogeochemical butterfly effect starts at the headwaters of rivers, under the toes of melting glaciers and permafrost. Glaciers and permafrost contain very old, unique nutrients, metals, and carbon when compared to surrounding areas. As temperatures persistently rise in high-latitude areas due to climate change, these glaciers and permafrost release these unique compounds, seeding downstream areas with an abundance of once-rare nutrients, including metals like cobalt. Though the rivers and oceans, these compounds are transported thousands of miles to different ecosystems capable of consuming them, leading to massive growth events throughout an entire food chain.
With Bundy et al’s work, even the tiny amounts of cobalt in the Arctic Ocean can now be quantified and understood. While this metal might be rare, it’s of great importance in the ocean ecosystem. The effects of climate change, however, can be found even here. The melting permafrost and glaciers of the Arctic have far-reaching, global implications as they release nutrients and minerals into major oceans, capable of changing whole ecosystems.
*
55 pmol L-1 is a measurement taken in Bundy et al. for the deep Arctic.
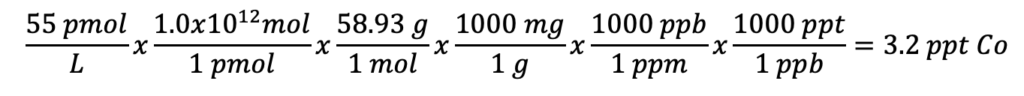
1 ppt is approximately equivalent of one grain of sand in an Olympic-sized swimming pool. 3.2 ppt is approximately equivalent to ~3 grains of sand per on Olympic swimming pool, therefore you would have a “better chance” of finding 5 grains of sand per Olympic swimming pool.
Unveiling the Mysterious Patterns of Arctic Cobalt by Jessica Buser-Young is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.

